Abirapro (Абиратерон 250mg)
Abirapro представляет собой лекарственное средство, которое широко применяется в онкологической практике в качестве препарата выбора для лечения метастазирующего рака предстательной железы.
Abirapro представляет собой лекарственное средство, которое широко применяется в онкологической практике в качестве препарата выбора для лечения метастазирующего рака предстательной железы. Имеет минимум побочных эффектов в сравнении с другими препаратами для лечения онкопатологий.
Состав и форма выпуска Абирапро
Является дженериком или полным аналогом препарата ZYTIGA. Производственные мощности фармацевтической компании находятся в Индии.
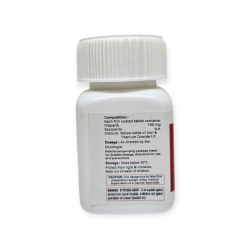
Действующим веществом выступает Абиратерон ацетат в максимально эффективной дозировке 250 мг.
Абиратерон ацетат синтетическим веществом, подавляющим выработку гормонов андрогенов, что позволяет эффективно замедлять рост новообразования.
В одной упаковке Абирапро 120 таблеток, предназначенных только для перорального приема.
Показания к применению Abirapro
Показанием к применению Abirapro является метастатический рак предстательной железы, который признан неоперабельным и кастрационно резистентным. Данное лекарственное средство применяют, в большинстве клинических случаев, в составе комплексной терапии, включающей:
- преднизолон;
- химиотерапию, включающую доцетаксел;
- другие препараты, замедляющие рост опухоли и угнетающими иммунный ответ.
Применять только по предписанию лечащего врача, после полного обследования пациента.
Противопоказания к применению Abirapro
Согласно официальной инструкции по применению, Abirapro не применяют для терапии:
- детей, не достигших возрастного порога 18 лет;
- пациентам с серьезными сопутствующими заболеваниями печени или почек;
- пациентов, страдающих от злокачественной гипертонии различной этиологии.
Не применяют для лечения женщин с другими формами патологических новообразований. С осторожностью принимают при сахарном диабете.
Возможные побочные реакции Абирапро
В ходе приема Абирапро, с различной частотой и интенсивностью, были отмечены следующие побочные реакции:
- боль в загрудинной области;
- повышение артериального давления;
- мигрень;
- головные боли различной локализации;
- зуд или жжение во время акта мочеиспускания;
- повышение уровня сахара в крови, постоянное чувство голода или жажды;
- тошнота, рвота и другие явления общей интоксикации;
- потеря веса, вплоть до анорексии;
- носовые кровотечения;
- аллергические реакции;
- боли в суставах;
- в отдельных случаях – сильный жар.
Наличие побочных реакций предполагает постоянный контроль артериального давления, а также уровня сахара в крови и других лабораторных показателей в соответствии с назначениями ведущего специалиста.
Все эти побочные эффекты могут встречаться в различных комбинациях.
В целом, препарат переносится пациентами достаточно хорошо,
Режим дозирования Abirapro
Эффективная схема лечения, предложенная специалистами представляет собой прием 1 грамма (4 таблетки Abirapro) единоразово в течение 24 часов.
Не принимать во время еды!
Таблетки принимают за 1 час до еды или через 2 часа после еды, обильно запивая питьевой водой.
Только для перорального приема.
Схема лечения назначается индивидуально для каждого пациента лечащим врачом в зависимости от имеющихся показателей , распространенности патологического процесса и некоторых других параметров.
Особенности применения Абирапро
Прием Абирапро может влиять на способность управлять транспортными средствами и механизмами, а потому от вождения автомобиля или работы с механизмами следует отказаться.
При любых изменениях общего соматического состояния больного, о них следует сообщить о них лечащему врачу.
Не принимать по окончанию срока годности.
Хранить в местах, недоступных для детей или домашних животных. Срок годности составляет 2 года.
| Фирменное наименование | Abirapro |
| Действующее вещество | Абиратерон 250mg |
| Упаковка | 120 Таблеток |
| Форма продукта | Таблетки |
Пока нет комментариев