Bevacirel 400mg (Bevacizumab)
Препарат Bevacirel 400 mg содержит Бевацизумаб — моноклональное антитело, направленное против фактора роста эндотелия сосудов (VEGF). Он блокирует образование новых кровеносных сосудов, питающих опухоль, тем самым «голодая» её. Назначается при колоректальном раке, немелкоклеточном раке лёгкого, раке яичников, глиобластоме и других злокачественных новообразованиях. Применяется внутривенно в составе комбинированной химиотерапии. Требует мониторинга артериального давления и контроля за риском кровотечений и тромбозов.
Препарат Bevacirel 400 mg содержит Бевацизумаб — рекомбинантное человеческое моноклональное антитело, которое избирательно связывается с VEGF (фактором роста эндотелия сосудов) и нейтрализует его биологическую активность. Это подавляет ангиогенез — формирование новых сосудов, необходимых для роста и метастазирования опухоли.
ПОКАЗАНИЯ
— Метастатический колоректальный рак (в комбинации с химиотерапией)
— Немелкоклеточный рак лёгкого нелегкооперабельный, локально-распространённый или метастатический
— Эпителиальный рак яичников, фаллопиевых труб или брюшины
— Глиобластома у взрослых при первичном или рецидивирующем заболевании
ВАЖНО
Бевацизумаб может вызывать тяжёлые побочные эффекты: носовые и желудочно-кишечные кровотечения, перфорацию кишечника, артериальные и венозные тромбозы, гипертензию и протеинурию. Противопоказан при активном кровотечении, тромбозах в анамнезе и непереносимости компонентов.
РЕЖИМ ДОЗИРОВАНИЯ
— При колоректальном раке: 5 мг/кг каждые 2 недели или 7,5 мг/кг каждые 3 недели
— При раке лёгкого: 15 мг/кг каждые 3 недели
— Вводится внутривенно в виде инфузии (30–90 минут) в онкологическом дневном стационаре
ПРОТИВОПОКАЗАНИЯ
— Гиперчувствительность к бевацизумабу
— Активное кровотечение или риск перфорации ЖКТ
— Тяжёлая артериальная гипертензия (неконтролируемая)
— Недавний инсульт или ИМ (в течение 6 месяцев)
ПОБОЧНЫЕ ЭФФЕКТЫ
Часто: гипертензия, утомляемость, головная боль, протеинурия, тошнота.
Серьёзно: геморрагия, перфорация кишечника, тромбоэмболия, задержка заживления ран, гипертонический криз.
ОСОБЫЕ УКАЗАНИЯ
Перед операцией терапию приостанавливают минимум за 4–6 недель. Избегать беременности — препарат вызывает повреждение плода. При появлении внезапной боли в животе, крови в кале или моче — срочно обратиться к врачу.
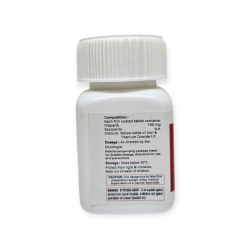
УСЛОВИЯ ХРАНЕНИЯ
Хранить в холодильнике при 2–8°C, не замораживать, оберегать от света. Не встряхивать.
| Фирменное наименование | Bevacirel 400 mg |
| Действующее вещество | Бевацизумаб |
Пока нет комментариев