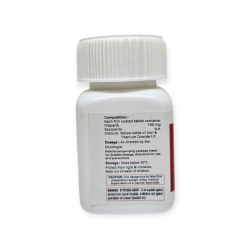
Thalitero 100mg (Thalidomide)
DESCRIPTION
Thalidomide is sold under brand name Thalitero .
Thalitero is a sedative drug discovered at the end of the 50s, which caused a worldwide tragedy. The drug has been prescribed to many pregnant women to relieve pregnancy nausea.
Thalitero is a prescription drug which is used under supervision of medical oncologist.
INDICATION
Thalitero isan antitumor medication
Used for the treatment in interaction with dexamethasone of patients with newly diagnosed multiple myeloma and been investigation for indication in various cancer condition like renal cell carcinoma, glioblastoma multiforme and Waldenström’s macroglobulinemia
Used for the treatment of skin lesions along with leprosy
MECHANISM OF ACTION
Thalidomide is known to have numerous effects on the immune system (immunomodulatory agent), which may provide to its therapeutic effect. Thalidomide may also change the production and activity of cytokines (growth factors) containing in the growth and survival of certain cancer cells. There may be an effect on the genes which direct the cell's development and activity specific for those analogues to cytokines (growth factors), apoptosis (cell death), and metabolism.
PHARMACOKINETICS
Peak plasma concentration is 2.9 to 5.7 hours.
The drug Thalitero has Plasma protein binding is 55% and 66% for the (+) R and (-) S enantiomers, respectively.
The drug Thalitero Metabolized hepatically by enzymes of the cytochrome P450 enzyme system.
Excreted in the urine as unchanged drug is less than 0.7% of the dose.
Half-life of thalidomide is 5 to 7 hours.
DOSAGE MANAGEMENT
The prescribed dose for Multiple Myeloma is 200mg PO qHS in 28-day cycles
Dexamethasone 40mg should administer on days 1-4,9-12,17-20 of 28 days.
The startingprescribed dose for Erythema Nodosum leprosumis 100-300 mg PO qHS.
If moreserious reaction then may initial at 400mg. enhance until active signsdiminish, at least 2 weeks then titrate down by 50 mg increment q 2-4 weeks.
PRECAUTION
• When treatment with Thalitero then Sexually mature women who have not go through a hysterectomy or who have not been postmenopausal for at least 24 consecutive months are treated to be women of childbearing potential
• Starting before Thalitero treatment, make sure to inform doctor about any other medications you are taking (containing prescription, over-the-counter, vitamins, herbal remedies, etc.). do not use aspirin, or products contains aspirin until the doctor especially permits this.
• Avoid administer drugs which may cause drowsiness without first consulting your health care provider. Also avoid alcohol.
• In case of both men and women use contraceptives, and do not get pregnantwhile taking Oncothal. severe, life-threatening birth defects may have resulted with even one 50 mg dose. Methods of contraception has two types, such as latex condoms and spermicides, are needed.
• While taking this Thalitero therapy avoid breast feed.
SIDE EFFECTS
Thalitero observes following side effects as follows :
• Thrombocytopenia
• Increased HIV viral load
• Bradycardia
• Seizures
• Tumor Lysis
• Hypersensitivity
• Asthenia
• Xerostomia
• Flatulence
• Neck rigidity
• Teratogenicity
• Somnolence
• Leukopenia
• Increased bilirubin
• Venous and arterial thromboembolism
• Increased Mortality
• Drowsiness and somnolence
• Peripheral neuropathy
• Dizziness and orthostatic hypotension
• Neutropenia
DRUG INTERACTION
Co administration of Thalitero with opioids, antihistamines, antipsychotics, anti-anxiety agents, other CNS depressants may lead to additive sedative effect.
Combination of Thalitero with cardiovascular medication will cause an additive bradycardic effect and should be used with caution.
Combination with bortezomib, amiodarone, cisplatin, docetaxel, paclitaxel, vincristine etc will cause peripheral neuropathy and should be used with caution.
When combination of hormonal contraceptives further increases the risk of thromboembolism with Thalitero is Unknown condition.
CONTRAINDICATION
Hypersentivity
Pregnancy - the drug cause risk to fetal
PREGNANCY
Pregnancy category X
Avoid Thalitero administer to a pregnant woman or a woman who aims to become pregnant.
Thalitero may cause fetal harm when administer to a pregnant woman.
If a woman becomes pregnant during Thalitero therapy, the drug must be stop immediately, and the woman given correct counselling.
LACTATION
Excretion of drug Thalitero in human milk and effects of drug on breastfed infants is unknown.
Avoid breastfeeding during treatment with Thalitero .
STORAGE
Store at 20°C - 25°C Protect the drug from light
MISSED DOSE
If dose is failed to take, Patients must consult with medical practitioner and follow the instructions given by them.
Hence missed dose must be avoid and follow the regular dosing schedule.
Пока нет комментариев