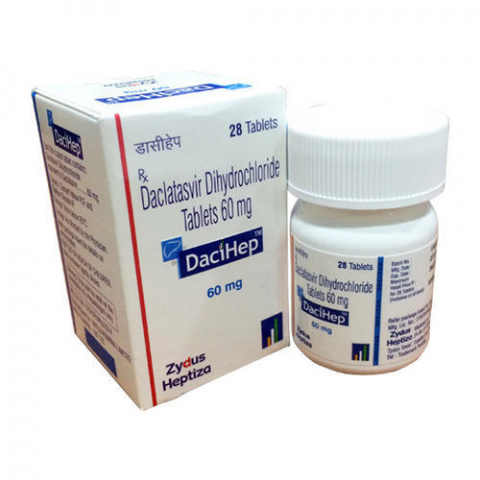
Dacihep (Daclatasvir)
Dacihep is indicated for the treatment of chronic hepatitis C viral infection by inhibiting the replication of hepatitis C virus.
DESCRIPTION
A virus which causes severe life threatening conditions is known as Hepatitis C virus that is a RNA virus, Chronic hepatitis Cirrhosis Hepatocellular carcinoma Dacihep is an anti-viral medication, which is available in tablet form. Dacihep perform its action by inhibiting the non structural 5A protein region of hepatitis C virus Dacihep is not monotherapy medicine; it should be combined with other anti-viral drugs for treating chronic hepatitis C infection caused by virus Dacihep is available in the strength of 30mg, 60mg, and 90mg
INDICATION
Dacihep is indicated for the treatment of chronic hepatitis C viral infection by inhibiting the replication of hepatitis C virus. Dacihep is indicated for the treatment of genotype III advanced hepatitis C virus in adults without cirrhosis. Normally Dacihep tablets are not used as single agent, it may combined with other anti-viral medicines like Sofosbuvir, interferon and ribavirin
MECHANISM OF ACTION
Dacihep is a NS5A inhibitors, NS5A protein is required for viral multiplication This NS5A inhibitor has efficiency to incorporate with HCV RNA. There are two essential mechanism in RNA proliferation which is based on phosphorylated state Cis acting function of phosphorylated NS5A: interfering HCV replication complex Trans acting function: regulate HCV assembly and infectious particle production Dacihep will disrupt hyperphosphorylated NS5A protein and mediated with newly formed RNA viral Dacihep involved in blockade of intercellular viral RNA synthesis and colony of virion
ADME PROPERTIES
ABSORPTION
The peak plasma concentration of Dacihep occurs within 2 hours. The mean bioavailability of Dacihep is 67%
DISTRIBUTION
The volume of distribution occurs at 47L. The human plasma protein bound with Dacihep with the range of 99%
METABOLISM
Dacihep a CYP3A substrates, it is dominantly metabolized by CYP3A4 isoenzymes High portion of drug in plasma is in unchanged mode nearly 97%
ELIMINATION
The route of elimination of metabolites; Elimination of drug via Bile: 88%; feces 53% (unchanged form) and 6.6% in unchanged form through urine The mean terminal half life period of Dacihep occurs from the range of almost 12 to 15 hours
DOSAGE MANAGEMENT
DOSAGE AND ADMINISTRATION OF DACLAHEP
Before initiating the therapy; NS5A resistance testing in HCV genotype 1a infected patients with cirrhosis has been evaluated The usual dose of Dacihep is 60mg as a single dose Recommended dosage regimens:
GENOTYPE I
Without cirrhosis: Dacihep + Sofosbuvir for 12 weeks Compensated cirrhosis (child Pugh A): Dacihep + Sofosbuvir for 12 weeks Decompensated cirrhosis (child Pugh B or C): Dacihep + Sofosbuvir + ribavirin for 12 weeks
GENOTYPE III
Without cirrhosis: Dacihep with Sofosbuvir for 12 weeks Compensated or decompensated cirrhosis: Dacihep with Sofosbuvir + ribavirin for 12 weeks
DOSE ADJUSTMENTS
While concomitant with strong CYP3A inhibitors: 30mg of Dacihep once a day Concomitant with moderate CYP3A inducers & nevirapine: 90mg of Dacihep as single dose With strong CYP3A inducers: contraindicated Dacihep should be administered with or without food
SIDE EFFECTS
While using the drug Some common side effects occurred as follows:: Chest pain, dizziness Irregular heart beat Unusual tiredness More common: Headache, nausea Pharmacological effects:
OTHER
Pyrexia , hot flush, loss of weight, Fatigue, asthenia, influenza like symptoms
NERVE
Headache, migraine, somnolence
DERMATOLOGICAL
Rashes, Pruritus, dry skin, alopecia, and Erythema multiforme
PSYCHIATRIC
Insomnia, irritability, depression, anxiety
BLOOD
Anemia, neutropenia, thrombocytopenia, esinophillia
RESPIRATORY
Cough, Nasopharyngitis, dyspnea, nasal congestion, upper respiratory infection
GIT
Gastro esophageal reflux, vomiting, Abdominal pain, elevated lipase, flatulence
MUSCLE
Myalgia, arthralgia, back pain
METABOLIC
Loss of appetite
CVS
Bradycardia, heart block, cardiac arrhythmias
LIVER
Increased AST & ALT, Hyperbilirubinaemia, hepatitis B reactivation
GENITOURINARY
Urinary tract infection
OCULAR
Dry eye
PRECAUTION & WARNING
There is a risk of adverse effects due to concurrent use of Dacihep with some other drugs may causes; loss of therapeutic effect serious life threatening conditions symptomatic bradycardia while using with amiodarone risk related to ribavirin during combination hepatitis b reactivation
DRUG INTERACTION
Concurrent use of Dacihep with some drugs likes; Amiodarone: serious bradycardia occurs Aprepitant: increase the serum concentration of CYP3A4 substrates Strong inducers of CYP3A causes decreased therapeutic effect of Dacihep Dacihep is an inhibitor of P-glycoprotein transporter, organic anion transporting polypeptide and breast cancer resistance protein; taking Dacihep , may elevate exposure to these drugs, which prolong the therapeutic effect Proteases inhibitors: increase effect of concentration of Dacihep Efavirenz, etravirine & nevirapine: decrease the effect of concentration of Dacihep Strong CYP3A inhibitors: increase the effect of concentration of Dacihep Interaction with HMG CoA reductase will increase the effect of concentration of these lipid lowering drugs.
CONTRAINDICATION
Concurrent use of CYP3A inducers strong with Dacihep , contraindicated to the patients Dacihep with ribavirin contraindicated to pregnancy condition Hypersensitivity reaction occur
PREGNANCY & LACTATION
During monotherapy: pregnancy category of Dacihep B; it is safe while using alone but under supervision of medical practitioner advice While ribavirin combination, the pregnancy categories with ribavirin X; causes severe ill effects like fetal harm even to death During monotherapy Breast feeding is safe but in combination with ribavirin breast feeding is not recommended
STORAGE
Dacihep tablets are stored at 25°C (77°F); excursion between 15°C and 30°C (59°F and 86°F) Keep away from heat, moisture and heat
MISSED DOSE
Dacihep is a prescription medicine; it should be used only under the guaindance of doctor Do not self medicate If patient fail to take the dose of Dacihep tablet, must consult with physician and take the missed dose within the time Or the missed dose should be leave and continue the regular dosing schedule Do not take overdose.
| Brand name | Dacihep |
| Active substance | Daclatasvir 60mg |
| Packaging | 28 Tablets |
| Product form | Tablet |
| Strength | 60mg |
There are no comments yet